


|
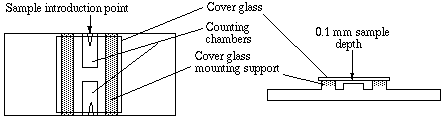 Diagramed above: Hemocytometer
Diagramed above: Hemocytometer< Pictured at left: Hemocytometer, RBC pipette, WBC pipette and Sahli pipette. |
|
RED CELL COUNT
The adjacent figure shows the rulings (improved Neubauer) which are inscribed on the counting chamber. The smallest squares in the large center square (where red cells are counted) have an area of 1/400 mm and are arranged in groups of 16. Each group of 16 squares is set off from the others by triple lines. The middle line is the one which actually defines the area of the squares adjacent to such a triple line. The inner line, therefore, is included within the area of the square. Erythrocytes in five of the 25 groups of 16 small squares are counted. These groups are the four corner groups and the one in the center. Count all the cells which touch the upper and left boundary lines (middle of the three lines) of the squares, but do not count those which touch the lower and the right boundary lines. A period of 2 to 3 minutes after filling allows the red cells to settle to the bottom of the chamber so that they will be in focus. Examine the chamber at 100X magnification for evenness of the red blood cell distribution. Turn the 40X objective in place; focus and count the cells in the designated squares. |
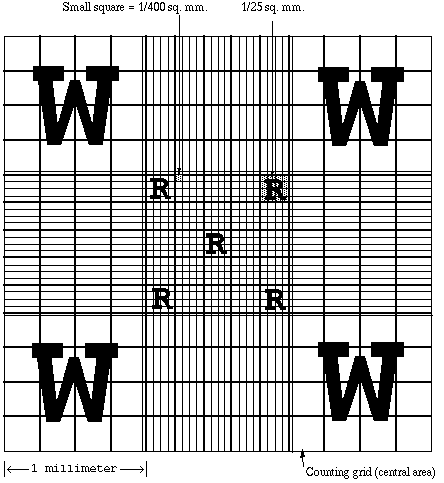 RED CELL COUNT - Count the five(5) small squares indicated by "R". Each of those squares contain 16 smaller squares. Use high power magnification (400X).
RED CELL COUNT - Count the five(5) small squares indicated by "R". Each of those squares contain 16 smaller squares. Use high power magnification (400X).
WHITE CELL COUNT - Count the four(4) large corner squares indicated by "W". Each of those squares contains 16 smaller squares the same size as one of the red cell squares. Use low power magnification (100X). |
|
Touch the end of a heparinized, capillary hematocrit tube to the edge of the blood sample and allow the tube to fill three-fourths full. Tilt the tube to leave both ends free of blood.
Seal one end of the tube with clay furnished with the tubes. Centrifuge for 5 min. at 10,000 RPM in a micro-hematocrit centrifuge, or for 30 min. at high speed in a regular single head centrifuge. After centrifugation, determine the hematocrit by measuring both the total height of blood and plasma and the height of the blood cell column (ignore the white buffy layer) to the nearest 1/2 mm. The hematocrit is calculated by: |

|

|
Draw 20 mm3 of blood into a Sahli pipette. Expel this blood into a test tube containing 5.0 ml of Drabkin's reagent. Rinse out the pipette twice with the Drabkin's reagent. Mix the contents using the vortex mixer. Determine the absorbance for this solution using the Spectronic 20 at 540 nm. Using the standard Hb curve, determine the Hb concentration of your blood.
< Spectronic-20 spectrophotometer |
| The WBC differential begins with preparation and staining of a blood smear. To prepare the smear place a small drop of blood on the surface of a clean microscope slide near the end; using a second |
| >

|
To stain the smears place the smears in Wright's stain for 3 minutes, transfer to water or phosphate buffer (pH 6.2) for 3 minutes and then briefly rinse with water. Place the slide in a vertical support and allow to air dry.
< Simple Wright's stain setup for staining blood smears Note the unstained and stained blood smears in the photo's foreground. |

|
Mount the stained blood smear on the microscope stage and examine at 400X magnification. Locate a region in the "feather" end of the smear where the RBCs are neither too difuse nor overlapping.
< Binocular microscope for blood smear exams 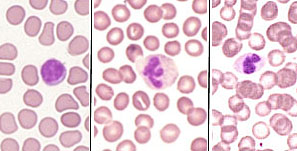 |
| Following the path illustrated at right, count the first 100 WBC encountered, classifying them by cell type and maturation. The tally is kept on a multichannel counter specifically designed for WBC differential counting. Do not include ruptured or incomplete cells (In the jargon of the trade, such cells are known as "skip-o-cytes"). |

|
|
|
| Search THIS ENTIRE WEB SITE for CLS technical, financial or other data >>> |
 E-mail
Irving E-mail
Irving |
This page is made entirely from recycled electrons. Any similarity between this and any other page may be completely intentional. Not tested on animals. Void where prohibited. Return to Irving's Home Page |